Decoding the Mitochondrial Genomic Reasons for Poor Bone Healing During Aging
Researchers uncover how mitochondrial DNA structures impair stem cells, offering new targets to improve bone healing in aging populations
CHENGDU, CHINA, April 22, 2026 /EINPresswire.com/ -- While aging is known to weaken bone repair, the underlying causes remain unclear. Researchers have now discovered that mitochondrial DNA structures called G-quadruplex accumulate in periosteal stem cells, disrupting energy production and driving cellular senescence. This shift impairs bone regeneration while increasing cartilage formation, a hallmark of poor healing. By identifying this mechanism, the study offers insights into potential targets for therapies to improve bone repair in older adults.Bone repair declines significantly with age, making fractures slower to heal and increasing the risk of long-term disability and reduced quality of life. While advances in regenerative medicine have improved treatment strategies, the underlying biological drivers of this decline have remained largely unclear. Emerging evidence now points to a previously overlooked factor: unusual DNA structures within mitochondria, the cell’s energy-producing centers. These structures, known as mitochondrial G-quadruplex (mtG4), accumulate over time and disrupt the function of periosteal stem cells—critical players responsible for effective bone regeneration.
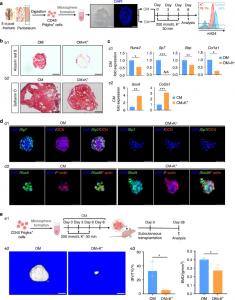
To address this challenge, a team of researchers led by Prof. Ling Ye, Prof. Fanyuan Yu and Dr. Feifei Li from State Key Laboratory of Oral Diseases & National Clinical Research Center for Oral Diseases, Sichuan University, China, investigated how mitochondrial DNA structures influence skeletal repair during aging. Using transgenic mouse models, stem cell-derived organoids, and advanced molecular analyses, the team systematically examined the role of mtG4 in periosteal mesenchymal stem cells. Their findings were published in Volume 14 of the journal Bone Research on April 07, 2026.
The researchers found that mtG4 structures accumulate specifically in periosteal stem cells during aging. This buildup disrupted normal mitochondrial gene expression, leading to reduced energy production, increased mitochondrial damage, and activation of cellular senescence pathways. As a result, these stem cells lost their ability to regenerate bone effectively and instead showed a shift toward cartilage formation—one of the defining features of impaired bone healing.
At the same time, experimental induction of mtG4 in young models reproduced aging-like defects, confirming a direct causal role. Cells with higher mtG4 levels exhibited reduced self-renewal, impaired osteogenesis, and increased inflammatory signaling, all of which contributed to poor skeletal repair outcomes. These findings establish a clear mechanistic link between mitochondrial DNA instability and age-related decline in bone regeneration.
“We identified mitochondrial G-quadruplex structures as a key upstream driver of stem cell dysfunction in aging bone, directly impairing the body’s ability to repair skeletal damage,” says Prof. Ye.
To further validate these findings, the team demonstrated that lowering mtG4 levels preserved stem cell function and improved regenerative potential. This highlights mtG4 not only as a biomarker of aging but also as a promising therapeutic target. The study suggests that interventions aimed at resolving or preventing mtG4 accumulation could restore the balance between bone and cartilage formation during healing.
“Our findings show that targeting these mitochondrial DNA structures could help restore stem cell function and open new avenues for improving bone healing in older individuals,” says Prof. Yu.
Beyond its mechanistic insights, the study highlights important implications for human health. In the short term, it may enable earlier identification of patients at risk of poor fracture healing and guide personalized treatment strategies. In the longer term, the discovery opens pathways for developing targeted senolytic therapies that selectively eliminate or rejuvenate dysfunctional cells without affecting healthy tissue.
The broader impact of this research extends across aging biology and regenerative medicine. By uncovering a mitochondrial DNA-based mechanism of stem cell decline, the study provides a new framework for understanding tissue degeneration in aging.
Ultimately, this work positions mitochondrial DNA structures as central regulators of skeletal aging, offering a promising route toward therapies that could restore bone healing capacity and reduce the burden of age-related fractures.
“Our study decodes the mitochondrial genomic reasons for poor bone repair during aging and offers insights into brand-new avenue towards enhancement of bone regeneration to reduce the huge burdens and death risks of fracture in elders,” says Dr. Li.
Reference
Titles of original paper: Periosteal mitochondria DNA structures drive aging-associated poor skeletal repair
Journal: Bone Research
DOI: https://doi.org/10.1038/s41413-026-00524-6
About Sichuan University
Sichuan University, located in Chengdu, China, is one of the country’s leading comprehensive universities with a strong emphasis on research, innovation, and interdisciplinary education. It is recognized for excellence in medical sciences, engineering, and life sciences, hosting nationally accredited laboratories and research centers. The university fosters international collaborations and supports cutting-edge scientific advancements addressing global health challenges. Through its commitment to academic excellence, Sichuan University continues to shape future leaders and innovators.
Website: https://en.scu.edu.cn/
About Professor Ling Ye from Sichuan University
Dr. Ling Ye is a Professor at the State Key Laboratory of Oral Diseases & National Clinical Research Center for Oral Diseases, Sichuan University, China. She currently serves as a Member of the Standing Committee of the Party Committee and Vice President of Sichuan University. Her research focuses on bone/tooth development and repair regeneration, and endodontics. She has published 40 SCI papers and has served as editor-in-chief or contributing author for 12 textbooks and monographs. In 2021, she was awarded the title of "Sichuan Province March 8th Red Flag Pacesetter" and the Baosteel Excellent Teacher Award.
About Prof. Fanyuan Yu from Sichuan University
Prof. Fanyuan Yu is a Professor at the State Key Laboratory of Oral Diseases & National Clinical Research Center for Oral Diseases, Sichuan University, China. His research focuses on stem cell fate determination and molecular mechanisms of hard tissue regeneration using transgenic lineage tracing technologies. He has authored 78 publications with 2,180 citations. His clinical and research work centers on endodontic diseases, regenerative therapies, and advancing innovative treatment strategies for bone repair.
About Dr. Feifei Li from Sichuan University
Dr. Feifei Li is affiliated to the State Key Laboratory of Oral Diseases & National Clinical Research Center for Oral Diseases, Sichuan University, China. Dr. Li has expertise in cell culture, polymerase chain reaction, Western blot analysis, immunofluorescence, cell line culture, and cell signaling. Dr. Li has authored 34 publications with 1,186 citations.
Funding Information
This work was supported by National Natural Science Foundation of China 82522021, 82571084, and 824B2023. Sichuan Province Science and Technology Program 2024ZYD0172, 2025ZNSFSC0754, 2025NSFJQ0071 (F.Y.), and 2024JDKXJ0001.
Yini Bao
Sichuan University
2885546461 ext.
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.